Manuscript Preparation
General Rules
Articles should be organized according to the ICMJE recommendations for the Conduct, Reporting, Editing, and Publication of Scholarly Work in Medical Journals updated in December 2019 (http://www.icmje.org/icmje-recommendations.pdf). CONSORT (www.consort-statement.org) for randomized trials, STROBE for observational studies (http://strobe-statement.org/), PRISMA for systematic reviews and meta-analyses (http://prisma-statement.org/), STARD for studies of diagnostic accuracy (http://www.equator-network.org/reporting-guidelines /stard/), ARRIVE for experimental animal studies (https://arriveguidelines.org/), and TREND for non- randomized public behavior (https://www.cdc.gov/trendstatement/) are other guidelines to help authors design their articles.
The upper limit of plagiarism determined by the editorial board for the journal is 20 percent. The filtering options in the plagiarism detection program are set to neglect references, quotes, and text sections of less than five words.
If there is an institution that supports the study, the last word of the article title should have an asterisk (*) and the information on the same page should be given as a footnote.
Manuscript Format
Title page
A separate title page should be submitted and this page should include:
- The full title of the manuscript, as well as a short title (running title) up to 50 characters,
- Name(s), affiliations, highest academic degree(s) and ORCID ID(s) of the author(s),
- Grant information and detailed information on the other sources of support,
- Nameandaddress,phone(includingthemobilephonenumber) number and email address of the corresponding author,
- Acknowledgment of the individuals who contributed to the preparation of the manuscript without fufilling the authorship criteria.
Abstract
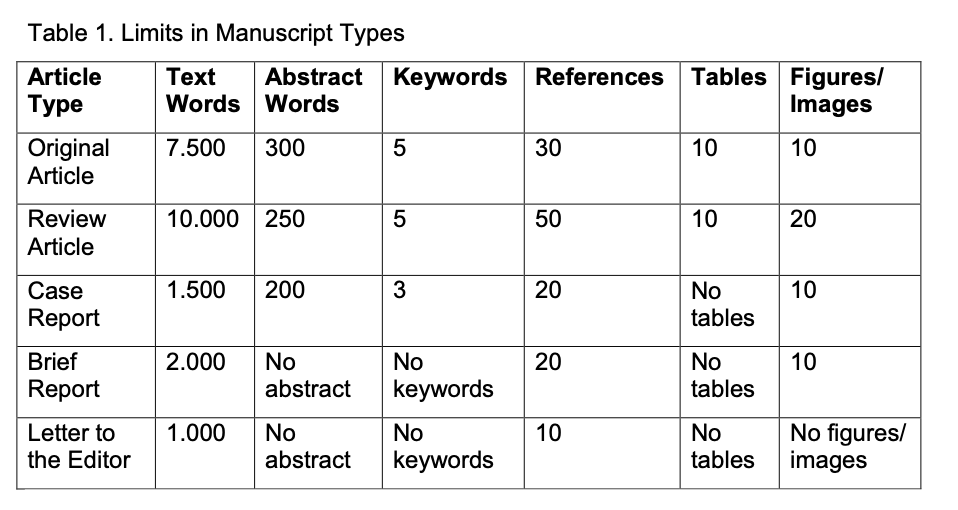
An English abstract should be submitted with all kind of manuscripts with the exception of Brief Reports and Letters to the Editor. The abstract of an Original Article should be constructed with subheadings (Objective, Methods, Results, and Conclusion). All acronyms and abbreviations used in the manuscript should be defined at first use, both in the abstract and in the main text. The abbreviation should be provided in parentheses following the definition. Please refer to Table 1 below for word count specifications.
Keywords
All manuscripts except Brief reports and Letters to the Editor must be accompanied by a minimum of three to a maximum of six keywords at the end of the abstract. Keywords should be selected from Medical Subject Headings (MeSH) of Index Medicus (https://www.nlm.nih.gov/mesh/MBrowser.html) Keywords will be used for subject indexing.
Manuscript Evaluation
Authors may send their articles, which are prepared in accordance with the below stated publishing and editorial principles, together with the “article presentation form” via e-mail to the provided addresses. Providing the permissions of all authors is obligatory. EJOH Editorial Board is authorized to decide whether or not to accept articles through international peer-reviews. Following the Section Editors’ preliminary reviews, the articles which are in accordance with the EJOH publication rules are sent to two reviewers determined by the Editor in Chief, for evaluation. In case of disagreement between the assigned reviewers, the manuscript is sent to a third reviewer. The articles which are sent back to the authors for further improvement, correction or revision should be edited accordingly and delivered back to the journal within one month at the latest. The results of corrections or revisions of the authors are re-examined by the reviewers and their decisions are reported to the editor. Manuscripts designated as appropriate for publication by the reviewers are sent to the statistical editor and if approved, the publication process begins. The articles which are found to be conflicting with this guideline, will be rejected and will not be issued.
Manuscript Types
The aim of the EURAS Journal of Health is to publish original research papers of qualified scientific value on health issues. Reviews to highlight relevant outstanding topics and developments, as well as case reports and brief reports or short notes to provoke research and discussion are also within the scope. The EURAS Journal of Health encourages and enables health professionals in the primary, secondary and tertiary health services to publish their research and reviews.
Original Article: Research article is the most important type of manuscript because it provides new information as a result of original research. The main text of original articles should be structured in detail with Introduction, Materials and Methods, Results, Discussion, and Conclusion subtitles and sections. Conclusions are supported by statistical analysis which is mainly necessary. Statistical analyses must be in accordance with international statistical reporting standards (Altman DG, Gore SM, Gardner MJ, Pocock SJ. Statistical guidelines for contributors to medical journals. Br Med J 1983:7;1489–93).
Information on statistical analyses should be supplied in a separate subheading under the Materials and Methods section and the statistical software that was used during the process must be included. Units should be prepared in accordance with the International System of Units (SI).
Limitations, drawbacks, and the shortcomings of original articles should be mentioned in the Discussion section before the conclusion paragraph.
Review Article: It is prepared by experts who have extensive knowledge in a particular field and whose intensive scientific background is translated into numeros publications with high impact potential. Experts should describe and evaluate the
current level of knowledge of a topic and guide future studies in the field. EJOH may also invite submissions from such authors. The main text should contain Introduction, Research Consequences, and Conclusion sections.
Case Report: Rare or challenging cases which are considered to be interesting and educative and those offering new therapies or revealing knowledge not included in the literature also are accepted for publication. Case Report should include the Introduction, Case Presentation, Discussion, and Conclusion sections.
Brief Report: Brief reports are similar to original research in that they build up with same structure such as content, format and guidelines but are designed for small scale scientific research outcomes that may contain preliminary data and initial findings that indicate need for further investigation. Brief reports are shorter than manuscripts and must contain significant data as in original research articles.
Letter to the Editor: It discusses important or neglected aspects of a previously published article for educative purposes. The text should be unstructured. Abstract, Keywords, and Tables, Figures, images, and other media should not be included. The manuscript that is being commented on must be properly cited within this manuscript.The manuscript that is being commented on must be properly cited within the Letter.
Writing Rules
- Page Layout: Paper size must be 17×24. Margins: top 3.5; down 2.5; left 2.5; right 2 cm
- Font: Times New Roman style and 11 pt. font size are used for the whole text. All article should be justified. Single line spacing should be used throughout the main text and between the paragraphs.
- Title: Bold capital letters in 14-pt must be used fort he main title. Subtitles should be written in bold and 11 pt,. After the title, author names, author ORCID numbers and e-mail addresses should be stated in 11 pt font size, with two lines of space.
- Abstract: Single paragraph in 11-pt, including subsections for Objective, Materials and Methods, Results and Conclusion sections are needed.
- Keywords should be in italic, bold type and 11 pt.
Figures and Images: Figures, graphics and photographs should be submitted as separate files (in TIFF or JPEG format). The files should not be embedded in a Word document or the main document. Images should not be labeled (a, b, c, etc.) to indicate figure subunits. When there are figure subunits, the subunits should not be merged to form a single image. Each subunit should be submitted separately through the submission system. As requested for the whole submission, the figures should also be blinded. Any information within the images that may indicate an individual or institution should be blinded. The minimum resolution of each figure should be 300 DPI. Figure legends can be supported by thick and thin arrows, arrowheads, stars, asterisks, and similar marks can be used on the images. Figure legends should be listed at the end of the main document. All submitted figures should be clear in resolution and large in size in order to prevent delayed evalutaion process.
When a drug, product, hardware, or software program is mentioned within the main text, product information, including the name of the product, the producer of the product, and city and the country of the company (including the state if in USA), should be provided in parentheses in the following format: “Discovery St PET/CT scanner (General Electric, Milwaukee, WI, USA)”
Tables
- The number of tables allowed for each type of manuscript is stated in Table 1.
- The tables should effectively display the information about desired levels of detail, so that the length of the text is also shortened. Each table should be printed on a separate page with double spacing.
- The table numbers should be given according to their order in the text, and each one should contain a short title. Authors should make explanations in footnotes, not in titles. All non-standard abbreviations should be explained in footnotes. The following symbols should be used for footnotes, respectively (*,†,‡,§,||,¶,**,††,‡‡).
- Statistical measurements of the variation (i.e. Standart error, standart deviation) should also be included. If data from another published or unpublished source are used, the cited source should be fully informed before its permission is obtained.
- Tables with a lot of data (backup) take up a lot of space and can only be suitable for electronic publications or they can be provided directly to the readers by the authors. In such a case, an appropriate expssion should be added to the text. Such tables should be submitted with the article for the evaluation by reviewer.
References: References are numbered and listed by their order of appearance in text; the text citation is followed by the appropriate reference number in parentheses. All references should be cited in text. While citing publications, priority should be given to the most up-to-date publications and accuracy of references is the responsibility of the authors. The DOI number should be provided ıf an ahead-of-print publication is cited. Journal titles should be abbreviated in accordance with the journal abbreviations in Index
Medicus/ MEDLINE/PubMed. All authors should be listed if there are six or fewer authors in the study. In case of six or more authorship, the first six authors should be listed followed by “et al.
Journal Article: Blasco V, Colavolpe JC, Antonini F, Zieleskiewicz L, Nafati C, Albanèse J, et al. Long-term outcome in kidney recipients from donors treated with hydroxyethylstarch 130/0.4 andhydroxyethylstarch 200/0.6. Br J Anaesth 2015;115(5):797-8.
Book Section: Philips SJ, Whisnant JP. Hypertension and Stroke. Editors: Laragh JH, Brenner BM. Hypertension pathophisiology, diagnosis, and management. 2nd ed. New York: Raven Press, 1995: 465–78.
Book with a Single Author: Jarvis C. Physical Examination and Health Assessment. 3rd ed. Philadelphia: W.B. Saunders Company; 2000.
Book with editor: Breedlove GK, Schorfheide AM. Adolescent pregnancy. Wieczorek RR, editor. 2nd ed. White Plains (NY): March of Dimes Education Services; 2001. p: 32-47.
Editor(s) as Author: Huizing EH, de Groot JAM, editors. Functional reconstructive nasal surgery. Stuttgart-New York: Thieme; 2003.
Conference Proceeding: Bengisson S. Sothemin BG. Enforcement of data protection, privacy and security in medical informatics. In: Lun KC, Degoulet P, Piemme TE, Rienhoff O, editors. MEDINFO 92. Proceedings of the 7th World Congress on Medical Informatics; 1992 Sept 6-10; Geneva, Switzerland.
Amsterdam: North-Holland; 1992. pp.1561-5.
Short Report: Cusick M, Chew EY, Hoogwerf B, Agrón E, Wu L, Lindley A, et al. Early Treatment Diabetic Retinopathy Study Research Group. Risk factors for renal replacement therapy in the Early Treatment Diabetic Retinopathy Study (ETDRS), Early Treatment Diabetic Retinopathy Study KidneyInt: 2004. Report No: 26.
Thesis: Borkowski MM. Infant sleep and feeding: a telephone survey of Hispanic Americans [dissertation]. Mount Pleasant (MI): Central Michigan University; 2002.
Manuscripts Accepted for Publication, Not Published Yet: Slots J. The microflora of black stain on human primary teeth. Scand J Dent Res. 1974.
Epub Ahead of Print Articles: Cai L, Yeh BM, Westphalen AC, Roberts JP, Wang ZJ. Adult living donor liver imaging. DiagnIntervRadiol. 2016 Feb 24. doi: 10.5152/dir.2016.15323. [Epub ahead of print].
Manuscripts Published in Electronic Format: Morse SS. Factors in the emergence of infectious diseases. Emerg Infect Dis (serial online) 1995 Jan-Mar (cited 1996 June 5): 1(1): (24 screens). Available from: URL: http:/www.cdc.gov/ncidodlElD/cid.htm
Webpage: Author. Title. Available at: URL. Accessed Access Date, Access Year.
Peer Review and Acceptance
Manuscripts are judged on the significance of the contribution to the literature, the quality of analysis and the clarity of presentation. Papers are expected to demonstrate originality and meaningful engagement with the global literature.
Except where otherwise stated, manuscripts are double-blind peer reviewed by anonymous reviewers in addition to the Editor. Final acceptance or rejection rests with the Editor-in-Chief, who reserves the right to refuse any material for publication.
Article Withdrawal
According to our publication policies, the author of the article has to cooperate with the Editor-in-Chief of the journal in withdrawal procedures, only if the reasons are compelling and unavoidable.
For withdrawal of a manuscript authors need to submit the petition containing his / her rationale to the editorial board in a printed signed form indicating that all authors have approved the withdrawal. Reasons for withdrawal should be stated clearly.
Authors must not assume that withdrawal process is completed until they have received appropriate notification to this effect from the editorial office. If the copyright of the article was transferred to the EJOH during the submission process, the author cannot send the work to another journal for evaluation unless the request for withdrawal of this work is approved.Cases of plagiarism, authorship disputes or fraudulent use of data will be handled in accordance with COPE guidelines.
Conflict of Interest
Authors must conceive any conflict of interest related to their submission. This statement must include any financial, personal or other relationships within three years of beginning the submitted work. When there is no such relationship, the authors must type “The authors declared they do not have anything to disclose regarding conflict of interest with respect to this manuscript.”
Authorship
The list of authors should accurately illustrate who contributed to the work and how. All those listed as authors should qualify for authorship according to the following criteria:
- 1. Have made substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data; and
- 2. Been involved in drafting the manuscript or revising it critically for important intellectual content; and
- 3. Given final approval of the version to be published. Each author should have participated sufficiently in the work to take public responsibility for appropriate portions of the content; and
- 4. Agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Contributions from anyone who does not meet the criteria for authorship should be listed, with permission from the contributor, in an Acknowledgments section (for example, to recognize contributions from people who provided technical help, collation of data, writing assistance, acquisition of funding, or a department chairperson who provided general support). Prior to submitting the article all authors should agree on the order in which their names will be listed in the manuscript.
The list of authors should accurately illustrate who contributed to the work and how. All those listed as authors should qualify for authorship according to the following criteria:
Authors must conceive any conflict of interest related to their submission. This statement must include any financial, personal or other relationships within three years of beginning the submitted work. When there is no such relationship, the authors must type “The authors declared they do not have anything to disclose regarding conflict of interest with respect to this manuscript.”